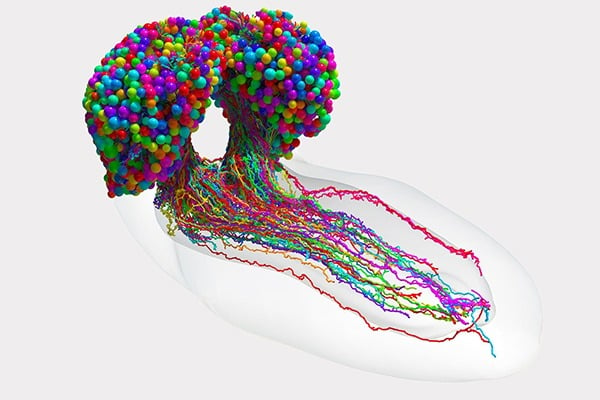
This team’s connectome of a baby fruit fly, Drosophila melanogaster larva, is the most complete as well as the most expansive map of an entire insect brain ever completed. It includes 3,016 neurons and every connection between them: 548,000.
“It’s been 50 years and this is the first brain connectome. It’s a flag in the sand that we can do this,” Vogelstein said. “Everything has been working up to this.”
Mapping whole brain is difficult and extremely time-consuming, even with the best modern technology. Getting a complete cellular-level picture of a brain requires slicing the brain into hundreds or thousands of individual tissue samples, all of which have to be imaged with electron microscopes before the painstaking process of reconstructing all those pieces, neuron by neuron, into a full, accurate portrait of a brain. It took more than a decade to do that with the baby fruit fly. The brain of a mouse is estimated to be a million times larger than that of a baby fruit fly, meaning the chance of mapping anything close to a human brain isn’t likely in the near future, maybe not even in our lifetimes.
The team purposely chose the fruit fly larva because, for an insect, the species shares much of its fundamental biology with humans, including a comparable genetic foundation. It also has rich learning and decision-making behaviors, making it a useful model organism in neuroscience. And for practical purposes, its relatively compact brain can be imaged and its circuits reconstructed within a reasonable time frame.
Even so, the work took the University of Cambridge and Johns Hopkins 12 years. The imaging alone took about a day per neuron.
Cambridge researchers created the high-resolution images of the brain and manually studied them to find individual neurons, rigorously tracing each one and linking their synaptic connections.
Cambridge handed off the data to Johns Hopkins, where the team spent more than three years using original code they created to analyze the brain’s connectivity. The Johns Hopkins team developed techniques to find groups of neurons based on shared connectivity patterns, and then analyzed how information could propagate through the brain.
In the end, the full team charted every neuron and every connection, and categorized each neuron by the role it plays in the brain. They found that the brain’s busiest circuits were those that led to and away from neurons of the learning center.
The methods Johns Hopkins developed are applicable to any brain connection project, and their code is available to whoever attempts to map an even larger animal brain, Vogelstein said, adding that despite the challenges, scientists are expected to take on the mouse, possibly within the next decade. Other teams are already working on a map of the adult fruit fly brain. Co-first author Benjamin Pedigo, a Johns Hopkins doctoral candidate in Biomedical Engineering, expects the team’s code could help reveal important comparisons between connections in the adult and larval brain. As connectomes are generated for more larva and from other related species, Pedigo expects their analysis techniques could lead to better understanding of variations in brain wiring.
The fruit fly larva work showed circuit features that were strikingly reminiscent of prominent and powerful machine learning architectures. The team expects continued study will reveal even more computational principles and potentially inspire new artificial intelligence systems.
“What we learned about code for fruit flies will have implications for the code for humans,” Vogelstein said. “That’s what we want to understand—how to write a program that leads to a human brain network.”