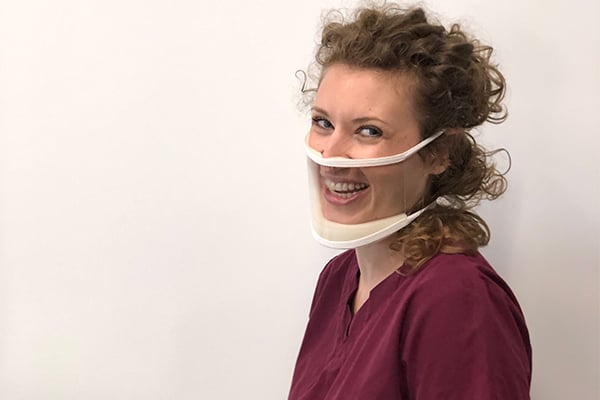
A transparent mask designed and created by a team of Johns Hopkins students and alumni is making a difference worldwide in the wake of the coronavirus pandemic. Eleven million of these masks have been sold to date, including to the United Kingdom National Health Service, which purchased 250,000 for frontline and social workers to better care for patients who use lip reading and facial expressions to communicate, such as those with hearing loss. Employees at Apple, Fortune 500 companies, and at state, local, national, and international governments are also using the masks.
The first fully see-through surgical mask cleared by the FDA, ClearMask was co-founded in 2016 by Allysa Dittmar, A&S ’14, SPH ’17, who is profoundly deaf. The inspiration for the design came after she experienced confusion and fear before a surgical procedure. She could not read the lips or see the facial expressions of the doctors, nurses, or anesthesiologists administering her care because the typical surgical masks were covering their faces.
Recognizing that many patients must experience the same situation, she and Aaron Hsu, A&S ’14, SPH ’15 spent the next several months coming up with a solution. When they found no alternative options on the market, they took matters into their own hands, researching market size and what was needed to develop a prototype of a transparent mask. Inez Lam, a doctoral student in biomedical engineering, and Elyse Heob, SPH ’18, Carey ’18, joined them as part of a Medical Entrepreneurship course at Johns Hopkins.
“Aaron pitched the idea to the class as a term-long project, and Elyse and I were both immediately drawn by Allysa’s compelling story and the potential impact of a transparent mask,” said Lam. “We believed in the idea and wanted to help it come to fruition.”